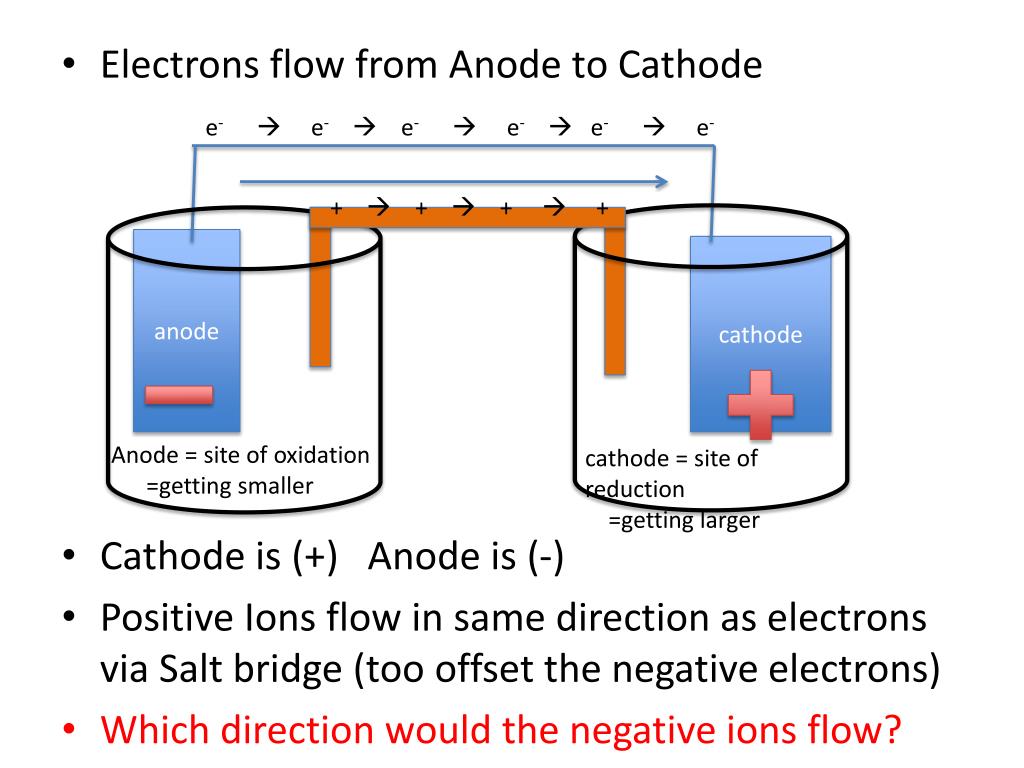
The anode is a positively charged electrode.It attracts cations or positive charges and is the source of electrons or an electron donor.The cathode is a negatively charged electrode.The concept of Anode is the same as it can act positive or negative both, but nature will depend upon the type of cell. In a discharging battery, it acts as positive whereas it acts negatively in another cell. A cathode is not positive or negative always. This Greek word means “way down” in which current flowing from positive to negative leaves a device in which anode lets the current passes in. The cathode was derived from the Greek word- “kathodos”. Since the electrons were discovered, a cathode is defined as negative, and an anode is defined as positive. Why are Cathode Negative and Anode Positive? Read More:- Difference between Galvanic Cells and Electrolytic Cells The electron would leave remain with an electron accumulation.Electrons would be reduced in the oxidized species.In Anode, there is oxidation response due to the following reasons: Electrons would be produced in the species of decreased ones.In Cathode, there is a reduction response due to the following reasons: Read More: Electrolytic Cells and Electrolysis It is an electrode where the electric current moves out. It is an electrode where the electric current moves into. Items to be plated with pure metal are attached and become part of the cathode in the solution.įollowing are the key differences between Anode and Cathode: When metal ions form a pure metal surface on the cathode, they are reduced. Electroplating Metal Cathode or Electrolysis The anode of the galvanic cell gives off electrons and returns from the circuit into the cell through the cathode. This positive pole is connected in order to allow the circuit to be completed. In a galvanic cell, the cathode is referred to as the positive pole. On the other hand, by studying the relative reducing power of two redox agents, the couple for making the more reducing species is more cathodic with respect to the more easily reduced reagent. Some results of reduction at the cathode are pure metal or hydrogen gas from metal ions. It is in an electrolytic cell where the negative polarity is applied in the cathode. The flow of electrons from the cathode interface to a species in solution is called the cathodic current. Learn More About: Electrochemical Cells Example of Cathode Hot Cathodes are heated with filament as it sends out electrons in thermionic emission.Cold Cathodes are not heated with filament as it sends out more electrons than the generation of electrons in thermionic emission only. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
